1. Studies & Services
1-1. Summary
|
|
|||
|---|---|---|---|
|
|
|||
|
Studies |
Site Users |
Patients |
Regulatory Approvals |
|
8,400+ |
182,000+ |
2,615,000+ |
350+ |
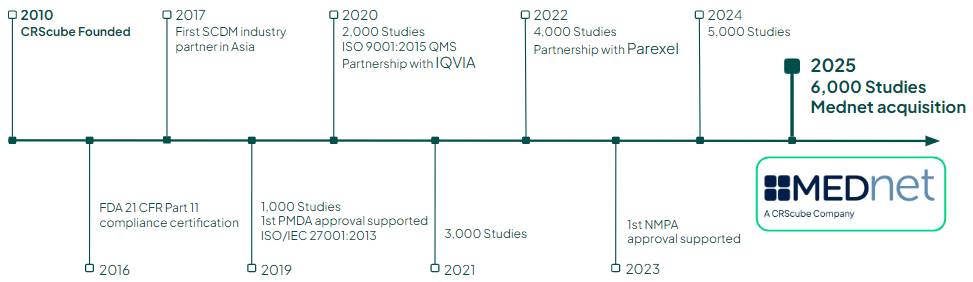
*combined with Mednet’s track record.
*All sections accept 1-1. Summary reflect CRScube’s independent track record
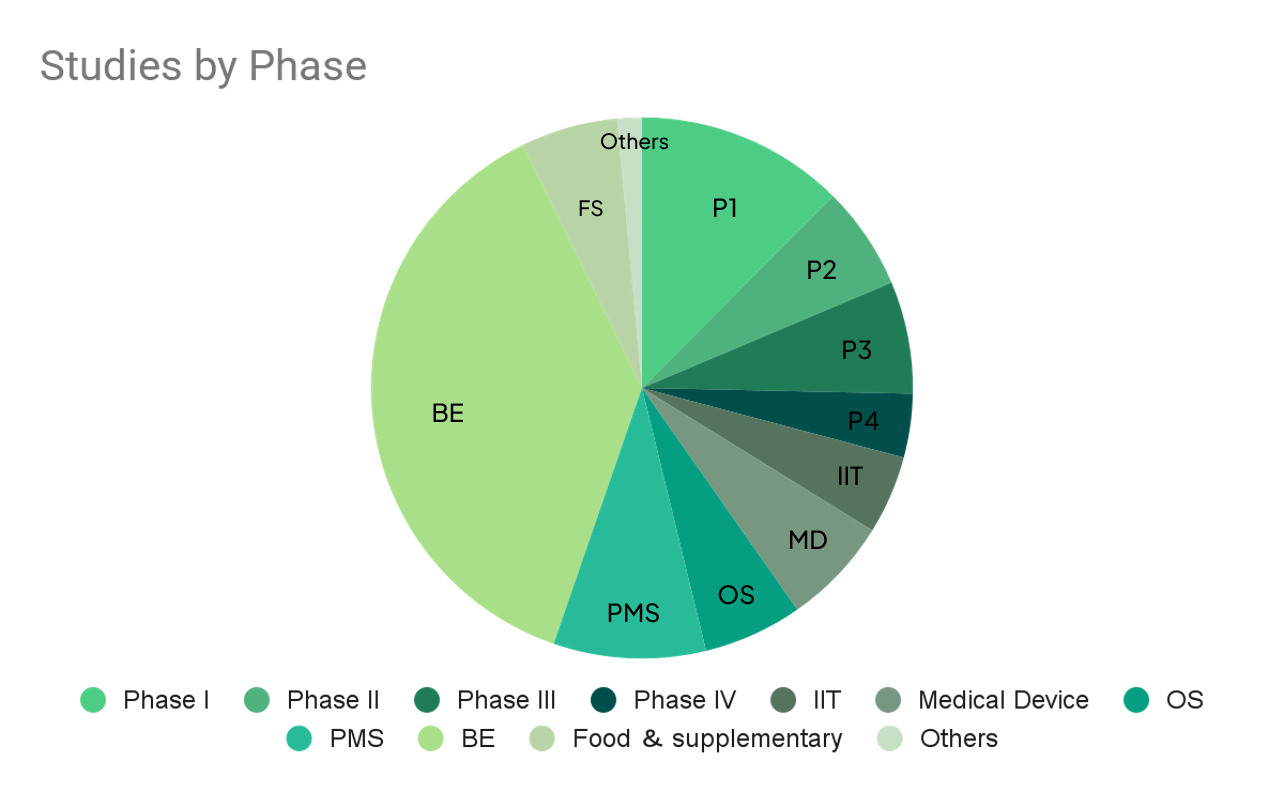
1-2. #No of Studies per Year and Phase
|
YEAR |
P1 |
P2 |
P3 |
P4 |
IIT |
MD |
PMS |
OS |
BE |
FS |
Other |
Total |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
2019 |
44 |
29 |
33 |
13 |
11 |
29 |
29 |
21 |
266 |
25 |
3 |
503 |
|
2020 |
74 |
67 |
37 |
21 |
41 |
35 |
49 |
40 |
317 |
61 |
11 |
753 |
|
2021 |
75 |
46 |
42 |
13 |
36 |
42 |
53 |
39 |
492 |
47 |
7 |
892 |
|
2022 |
88 |
32 |
40 |
17 |
38 |
50 |
55 |
33 |
347 |
65 |
14 |
779 |
|
2023 |
114 |
33 |
48 |
10 |
40 |
85 |
68 |
32 |
257 |
60 |
13 |
760 |
|
2024 |
176 |
43 |
61 |
17 |
38 |
74 |
71 |
42 |
238 |
75 |
16 |
851 |
|
2025 |
161 |
23 |
32 |
21 |
43 |
66 |
69 |
48 |
238 |
35 |
15 |
751 |
|
2026 (Ongoing) |
57 |
10 |
7 |
3 |
13 |
18 |
24 |
5 |
57 |
4 |
3 |
201 |
|
Total (Since 2010) |
897 |
325 |
411 |
244 |
301 |
410 |
583 |
379 |
2382 |
365 |
97 |
6394 |
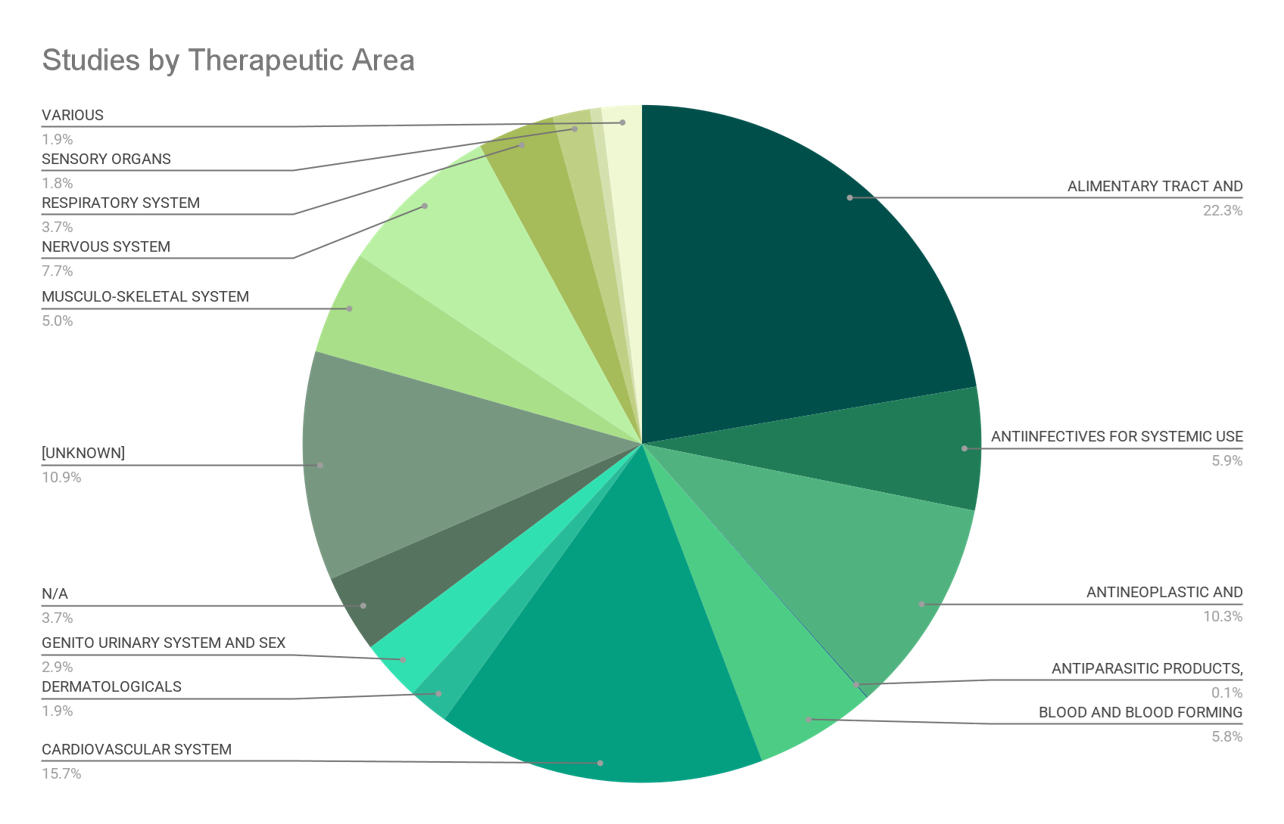
1-3. #No of Studies per Therapeutic Area and Phase
|
Therapeutic Areas (ATC Classification-based) |
P1 |
P2 |
P3 |
P4 |
MD |
IIT |
PMS |
OS |
BE |
FS |
Other |
Total |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
ALIMENTARY TRACT AND METABOLISM |
179 |
37 |
90 |
48 |
0 |
37 |
68 |
52 |
582 |
312 |
15 |
1420 |
|
ANTIINFECTIVES FOR SYSTEMIC USE |
48 |
35 |
38 |
17 |
0 |
8 |
67 |
22 |
133 |
0 |
7 |
375 |
|
ANTINEOPLASTIC AND IMMUNOMODULATING AGENTS |
152 |
81 |
22 |
32 |
0 |
93 |
141 |
55 |
61 |
4 |
16 |
657 |
|
ANTIPARASITIC PRODUCTS, INSECTICIDES AND REPELLENTS |
2 |
1 |
1 |
0 |
0 |
0 |
1 |
0 |
0 |
0 |
0 |
5 |
|
BLOOD AND BLOOD FORMING ORGANS |
61 |
22 |
18 |
13 |
2 |
26 |
21 |
42 |
155 |
0 |
11 |
371 |
|
CARDIOVASCULAR SYSTEM |
132 |
14 |
78 |
45 |
0 |
24 |
87 |
94 |
529 |
3 |
0 |
1006 |
|
DERMATOLOGICALS |
21 |
12 |
21 |
4 |
13 |
7 |
19 |
5 |
12 |
8 |
1 |
123 |
|
GENITO URINARY SYSTEM AND SEX HORMONES |
22 |
6 |
10 |
4 |
0 |
4 |
16 |
11 |
110 |
8 |
1 |
192 |
|
MUSCULO-SKELETAL SYSTEM |
39 |
21 |
26 |
16 |
5 |
9 |
35 |
11 |
142 |
5 |
7 |
316 |
|
NERVOUS SYSTEM |
28 |
21 |
17 |
30 |
2 |
18 |
42 |
29 |
285 |
5 |
3 |
480 |
|
RESPIRATORY SYSTEM |
14 |
10 |
25 |
5 |
0 |
5 |
29 |
14 |
126 |
3 |
3 |
234 |
|
SENSORY ORGANS |
19 |
23 |
26 |
11 |
0 |
3 |
16 |
10 |
3 |
2 |
2 |
115 |
|
SYSTEMIC HORMONAL PREPARATIONS, EXCL. SEX HORMONES AND INSULINS |
2 |
1 |
5 |
4 |
0 |
3 |
7 |
6 |
7 |
0 |
0 |
35 |
|
VARIOUS |
16 |
8 |
5 |
7 |
28 |
13 |
10 |
12 |
8 |
9 |
8 |
124 |
|
N/A |
10 |
5 |
6 |
2 |
175 |
22 |
9 |
5 |
10 |
1 |
10 |
255 |
|
[UNKNOWN] |
152 |
28 |
23 |
6 |
185 |
29 |
15 |
11 |
219 |
5 |
13 |
686 |
|
Therapeutic Areas (Indication-based) |
P1 |
P2 |
P3 |
P4 |
MD |
IIT |
PMS |
OS |
BE |
FS |
Other |
Total |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Cardiovascular & Circulatory |
132 |
14 |
78 |
45 |
0 |
24 |
87 |
94 |
529 |
3 |
0 |
1006 |
|
Dermatology |
21 |
12 |
21 |
4 |
13 |
7 |
19 |
5 |
12 |
8 |
1 |
123 |
|
Endocrinology & Metabolic |
121 |
15 |
41 |
23 |
0 |
17 |
43 |
30 |
209 |
0 |
2 |
501 |
|
Gastroenterology |
60 |
23 |
54 |
29 |
0 |
23 |
32 |
28 |
380 |
312 |
13 |
954 |
|
Immunology & Inflammation |
26 |
9 |
5 |
19 |
0 |
10 |
53 |
16 |
22 |
0 |
8 |
168 |
|
Infectious Diseases |
14 |
22 |
6 |
15 |
0 |
6 |
30 |
15 |
133 |
0 |
6 |
247 |
|
Mental Health & Behavioral |
18 |
10 |
5 |
14 |
0 |
8 |
13 |
16 |
147 |
2 |
2 |
235 |
|
Nephrology |
39 |
13 |
13 |
3 |
0 |
4 |
2 |
13 |
0 |
0 |
7 |
94 |
|
Neurology/Nervous System |
10 |
11 |
12 |
16 |
2 |
10 |
29 |
13 |
138 |
3 |
1 |
245 |
|
Oncology |
118 |
66 |
14 |
12 |
0 |
81 |
83 |
36 |
39 |
0 |
8 |
457 |
|
Ophthalmology |
18 |
23 |
26 |
11 |
0 |
3 |
16 |
10 |
3 |
2 |
1 |
113 |
|
Other / Undefined |
248 |
77 |
68 |
42 |
395 |
97 |
93 |
71 |
534 |
24 |
43 |
1692 |
|
Respiratory Diseases |
14 |
10 |
25 |
5 |
0 |
5 |
29 |
14 |
126 |
3 |
3 |
234 |
|
Vaccines & Immunotherapies |
36 |
14 |
33 |
2 |
0 |
2 |
38 |
7 |
0 |
0 |
1 |
133 |
|
Women’s Health & Gynecology |
22 |
6 |
10 |
4 |
0 |
4 |
16 |
11 |
110 |
8 |
1 |
192 |
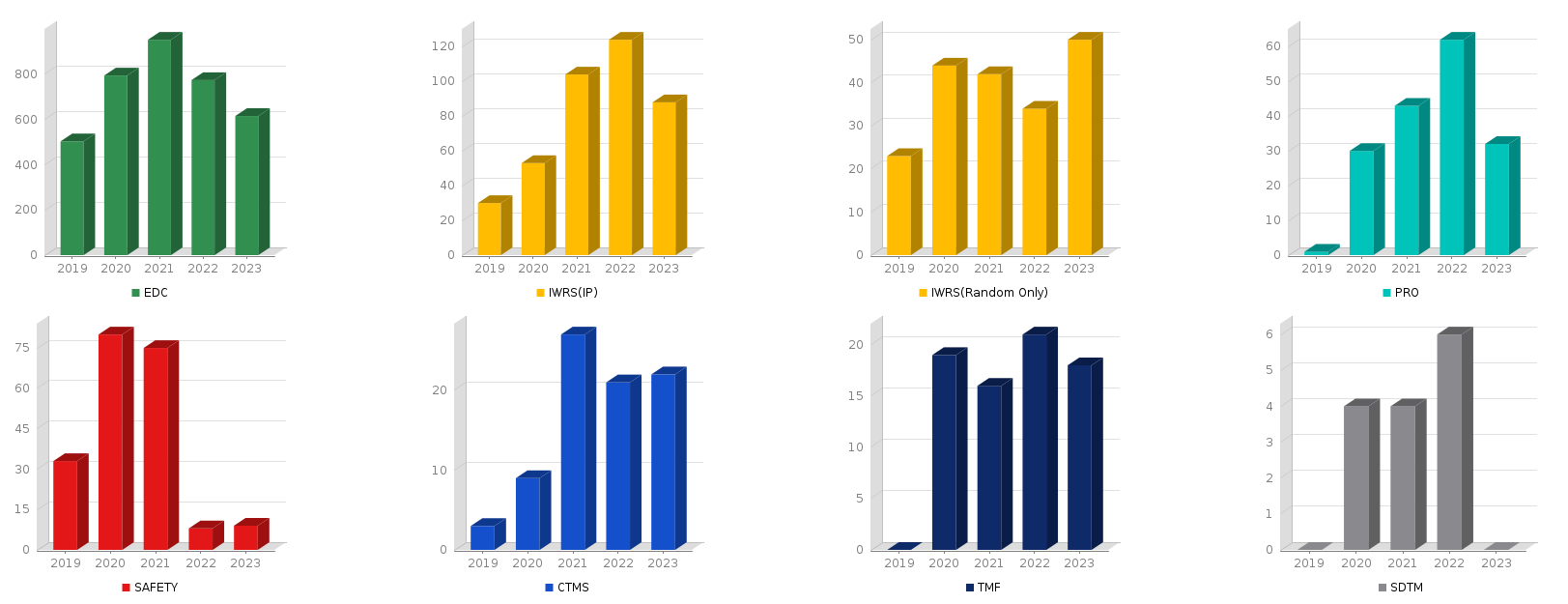
1-4. #No of Services per Year
|
YEAR |
EDC |
IWRS (IP) |
IWRS (Random Only) |
PRO |
cubeSAFETY |
CTMS |
TMF |
DM |
SDTM |
|---|---|---|---|---|---|---|---|---|---|
|
2019 |
503 |
30 |
23 |
1 |
41 |
3 |
. |
17 |
. |
|
2020 |
749 |
50 |
40 |
30 |
80 |
9 |
19 |
31 |
4 |
|
2021 |
891 |
96 |
42 |
40 |
77 |
30 |
16 |
31 |
4 |
|
2022 |
774 |
123 |
35 |
62 |
9 |
17 |
17 |
20 |
6 |
|
2023 |
736 |
112 |
58 |
46 |
12 |
29 |
25 |
18 |
. |
|
2024 |
842 |
137 |
56 |
49 |
15 |
23 |
17 |
9 |
1 |
|
Total |
4516 |
551 |
255 |
232 |
234 |
112 |
98 |
126 |
15 |
1-5. #No of DM Studies per Year
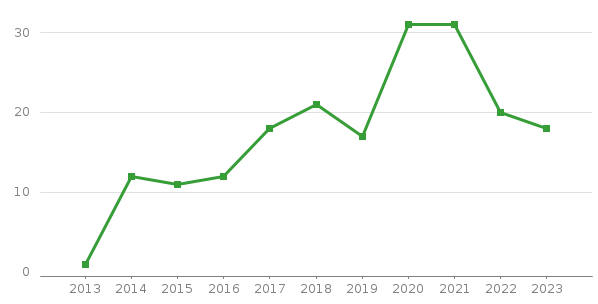
|
Year |
2013 |
2014 |
2015 |
2016 |
2017 |
2018 |
2019 |
2020 |
2021 |
2022 |
2023 |
2024 |
|
Total |
1 |
12 |
11 |
12 |
18 |
21 |
17 |
31 |
31 |
20 |
18 |
9 |
2. Global Experience
2-1. US
|
Sponsor |
Phase |
Indication |
Note |
|---|---|---|---|
|
P** (Biotech) |
II |
Oncology |
|
|
H** (Biotech) |
I/II |
LARS (Low Anterior Resection Syndrome) |
|
|
O** (Biotech) |
II |
COVID 19 |
Emergency Use Authorization expected in the 1st quarter of 2024 cubeCDMS + cubeSAFETY |
|
V** (Biotech) |
I |
Oncology |
|
|
V** (Biotech) |
II |
Oncology |
cubeCDMS + cubeSAFETY |
|
O** (Biotech) |
II |
Atopic Dermatitis |
|
|
N** (Biotech) |
I |
Oncology |
cubeCDMS + cubeIWRS |
2-2. EU
|
Sponsor |
Phase |
Indication |
Note |
|---|---|---|---|
|
S** (Pharmaceutical company) |
OS |
NA |
Bulgaria, Czechia, France, Poland, Romania, Russia, Ukraine |
|
M** (Biotech) |
III |
NA |
Russia |
|
D** (Biotech) |
Pivotal
|
Transrectal Prostate Biopsy |
Poland |
|
S** (Biotech) |
Pivotal
|
Prediabetes |
Hungary, Romania, Slovakia |
|
G** (Pharmaceutical company) |
III |
NA |
Poland |
|
P** (Biotech) |
II |
Diabetes |
Poland |
|
P** (Pharmaceutical company) |
I |
NA |
Poland |
|
P** (Pharmaceutical company) |
I&II |
Primary Progressive Multiple Sclerosis, PPMS |
Poland |
|
S**(Pharmaceutical company) |
II |
Ophthalmic/ocular Diseases |
Poland |
|
S**(Pharmaceutical company) |
III |
PGTCS |
Serbia, Macedonia |
|
C**(Pharmaceutical company) |
III |
Rheumatoid Arthritis |
Poland |
|
C**(Pharmaceutical company) |
III |
Plaque Psoriasis |
Poland |
|
N**(Pharmaceutical company) |
OS |
NA |
Poland |
2-3. Phase 3
|
Country |
Regulation Agency |
No of Studies |
|---|---|---|
|
Korea |
MFDS |
More than 30 per year |
|
Japan |
PMDA |
36 |
|
China |
NMPA |
12 |
|
Taiwan |
TFDA |
2 |
|
EU |
EMA |
6 |
2-4. Regulatory Body Inspection
In most regulatory authorities, conducting inspections solely for eClinical solutions is rare. Still, we have compiled the following summary in response to commonly asked questions from our clients. The parentheses next to the number of studies indicate the following categories:
-
Some eClinical solutions were included in the study approval inspection process.
-
Inspection on eClinical solutions.
|
Country |
Regulatory Agency |
No. of Studies |
Notes |
|---|---|---|---|
|
Korea |
MFDS |
>= 15 per year (1) |
|
|
Japan |
PMDA |
10 (1) |
|
|
China |
NMPA |
1 (1) 3 (2) |
(1) Small cell lung cancer. (2) NMPA inspection on eClinical solutions used in China. |
|
Rwanda |
Rwanda FDA |
1 (2) |
COVID-19 vaccine. |
|
Philippines |
Philippine FDA |
1 (2) |
COVID-19 vaccine. |
|
USA |
FDA |
|
|
|
Taiwan |
TFDA |
NA |
|
|
EU |
EMA |
NA |
|
2-5. Studies per Region and Phase
|
Region |
P1 |
P2 |
P3 |
P4 |
MD |
IIT |
MS |
OS |
BE |
FS |
Other |
Total |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Korea |
354 |
229 |
278 |
202 |
248 |
124 |
375 |
270 |
1857 |
259 |
572 |
4261 |
|
Japan |
51 |
45 |
28 |
3 |
12 |
82 |
36 |
15 |
22 |
2 |
21 |
303 |
|
China |
48 |
30 |
11 |
0 |
0 |
2 |
2 |
4 |
38 |
1 |
2 |
137 |
|
Taiwan |
1 |
2 |
0 |
0 |
1 |
0 |
1 |
4 |
0 |
0 |
1 |
9 |
|
US |
1 |
3 |
0 |
0 |
0 |
2 |
0 |
0 |
0 |
0 |
0 |
6 |
|
Europe |
1 |
0 |
1 |
0 |
1 |
0 |
0 |
7 |
0 |
0 |
1 |
10 |
|
ROW |
9 |
10 |
16 |
2 |
0 |
1 |
1 |
14 |
0 |
0 |
0 |
53 |